2013年第40卷第12期目录
| |
|
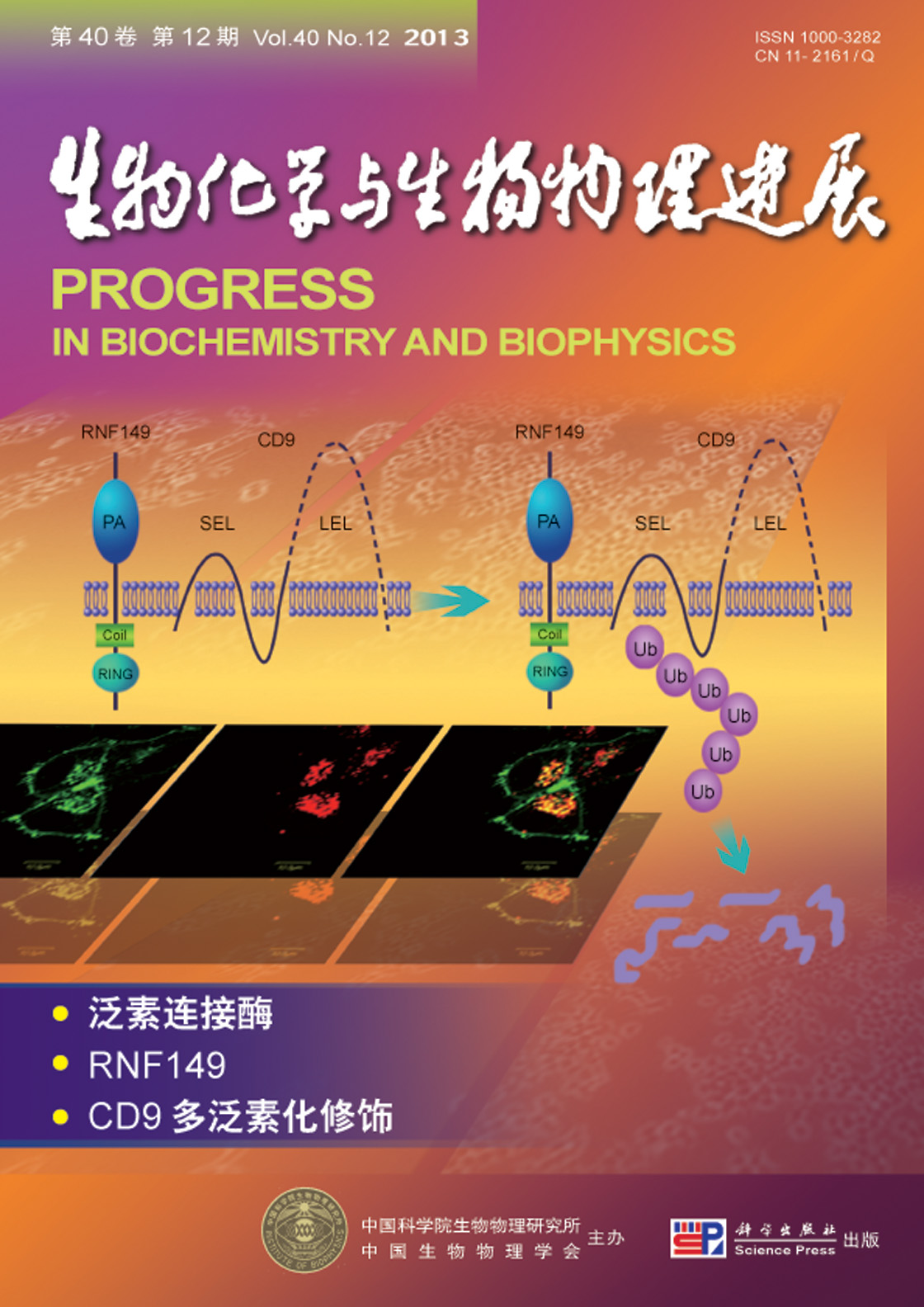
封面故事:泛素化修饰做为一种重要的蛋白质共价修饰,对于细胞正常生理活动的调控有重要作用.泛素连接酶对被泛素化修饰的蛋白质分子有特异识别作用,在蛋白质泛素化反应中发挥至关重要的作用.目前,关于膜蛋白类泛素连接酶的研究很少,此前的研究表明,属于膜蛋白类的泛素连接酶GRAIL家族蛋白,在免疫调控中具有重要的功能.李燕等的研究结果揭示了GRAIL 家族中功能未知的成员RNF149的细胞器定位及其与底物作用方式.RNF149定位于溶酶体,能够通过多泛素化修饰CD9来介导CD9的降解,并且通过调控CD9的量来影响细胞增殖.另外,CD9能被多泛素化修饰发生降解也属首次报道.
(李 燕,阮林浩,索塔林,王 鹏,唐 捷. RNF149通过泛素化介导的CD9降解调控细胞增殖,本期第1230~1238页)
Cover Story:Our research selected the RNF149, the novel ubiquitin ligase which owned the high identity to GRAIL and belonged to the typeⅠ transmembrane protein, as our object. By the confocal laser scanning microscope, it was demonstrated that RNF149 is located at lysosome and the RNF149 is co-located with CD9. The interactions between RNF149 and CD9 were demonstrated by immune co-precipitation.RNF149 polyubiquitinates CD9 via ubiquitin Lys-48. The HeLa cells were co-transfected with the same quantity of the CD9 plasmids and the gradient increase quantity of the RNF149 plasmids. We found that the exogenous quantity of CD9 was decreasing with the increased expression of the exogenous RNF149. In HEK293T cells, the knocking down RNF149 by shRNA led to the increase of the endogenous CD9. All these evidence suggested that CD9 maybe regulated by RNF149. In addition, the knocking down RNF149 by shRNA led to the inhibition of the cell proliferation in HEK293T cells. This phenomenon suggested that the RNF149 possibly could be considered as the regulatory factor of the cell proliferation.
|
综述与专论
研究报告
技术与方法
|
|